|
In a triple bond, there is one sigma and two pi bonds (e.g., -C≡C-). As in a double bond, we have one sigma and one pi (e.g., -C=C-). For atoms having multiple bonds, the first bond is sigma, and the remaining are pi. Two different atoms can only have one sigma bond between them. And the two carbons are bonded by an sp3-sp3 sigma bond. Each carbon bonds with three hydrogens by s-sp3 sigma bonds. The sigma bond is also formed with a hybrid orbital, for example, s-sp3, sp3-sp3, and so on.Īs an example, in ethane (H3C-CH3), the carbon atoms are sp3 hybridized. 
The greek symbol “σ” is used to denote them, and the electrons belonging to σ bonds are termed σ electrons.īesides the s-s overlap, we can have s-p, p-p, s-dz2, dz2-dz2, and so forth. Sigma bonds are stronger, stable, and are less likely to break over pi bonds during a chemical reaction. This makes sigma bonds the strongest covalent bond. In sigma bonds, the overlapping is head-to-head in other words, it is a direct overlap. The atoms are free to rotate along the bond axis. The line joining the two nuclei is the bond axis. But after the formation of the sigma bond, the probability of finding the bonding electrons is maximum between two nuclei of the atoms. In the individual atoms, the electron density is higher toward the center of the atom, i.e., near the nucleus. In the above diagram, the s-orbital of one atom overlaps with the s-orbital of the second. Consider the simplest case of the sigma bond, where a diatomic molecule is formed by sharing an s-orbital of each atom. In sigma bonds, each orbital of two atoms overlaps with each other head-to-head. 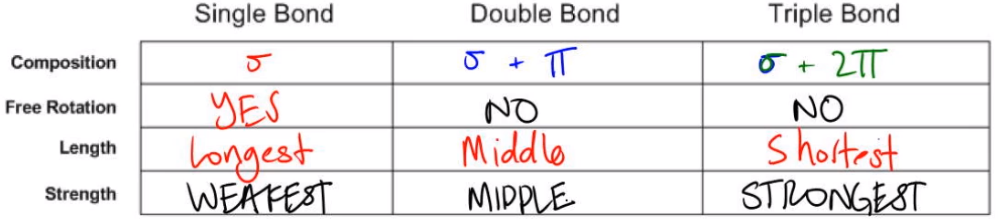
There are other types, too, such as delta bonds. Note: The covalent bond is not limited to sigma and pi bonds. And based on a type of overlap, we classify them as a sigma or pi bond. The resultant orbitals after overlapping are called molecular orbitals.ĭifferent orbitals make different types of overlap in different situations. The overlapping of orbitals can be virtually thought of as the sharing of space between two nuclei, where the probability of finding bonding electrons is maximum. 
During chemical bonding, the orbitals of two different atoms come close to each other. Atoms contain orbitals, and orbitals hold electrons. In a covalent bond, chemical bonding is achieved by sharing of electrons. Sigma and pi bonds are the most popular forms of covalent bonds.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
